Monkeypox Virus MPV Nucleic Acid Detection Kit Real Time PCR Method
|
Monkeypox Virus(MPV) Nucleic Acid Detection Kit (Real-Time PCR Method)››› World’s Fastest Covid Testing Kit! ››› Easy operation, Safe and accurate, Money-saving!!
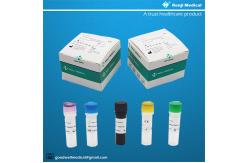
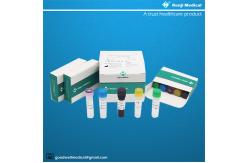
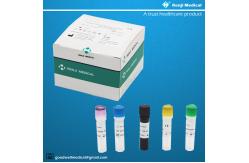
【PRODUCT NAME】 Monkeypox Virus (MPV) Nucleic Acid Detection Kit (Real-time PCR Method)
50 tests/kit & 200 tests/kit 【Intended Use】 This kit is used forthe detection of Monkeypox Virus(MPV) in serum
or lesion exudate samples by using real
【Test Principle】
【Components of the Diagnostic Kit】
Note: 1. Do not mix or exchange components from different kit lots. 2.The MPV Negative Control is sterile nuclease-free water and
Internal Control(IC) gene, and the MPV Positive Control is DNA in
vitro which contains target genes and Internal Control(IC) gene. 【Storage and Stability】 1. The diagnostic kit should be stored in a sealed pouch at -20 ± 5℃. The expiry date is 12 months. 2. Please refer to the date of manufacture and expiry date on the
outer package. 3. The reagent keep valid and stable within the expiry date if not
used. The kit should not be frozen and thawed more than 5 times.
【Applicable Instrument】
【Specimen Requirements】
2.Specimens can be extracted immediately or frozen at -20°C to -80°C.
3.Transportation of clinical specimens must comply with local regulations for the transport of etiologic agents.
【The Sample Result Judgment 】 2.If the test sample has no Ct or Ct > 38 in the FAM channels, and there is a typical S-type amplification curve in the Internal Control channel(Cy5), the Ctis ≤38.the sample canbe judgedtobeMPVnegative. 3.If the test sample have no typical S-type amplification curve (No Ct value) or Ct value> 38 is detected in the FAM, andCy5 channel does not have a typical amplification curve (No Ct value) or Ct value> 38, it means that there is a problem with the quality of the sample or a problem with the operation. If the result is invalid, you should find and eliminate the cause,collectthe sampleagain, andrepeatthe test(ifthe test resultis stillinvalid,please contact the company).
【Limitations of Detection Method】 2.False negative results may occur when the concentration of the detecte dnucleicacid in the test sampleis below the minimumde tection limit of this kit. 3.Improperhandlingofthetestedsampleduringcollection,transportation, storage, andprocessingcaneasily resultin DNA degradation and falsenegative results. 4.When samples are cross-contaminated during collection,transportation, storage, and rocessing,it is easy togetfalse positive results.
【Product Performance Index】
【Precations】 2.Negative andpositive controls shouldbe setfor each experiment. 3.All reagents in the kit should be fully thawed and mixed at room temperature and centrifuged immediately before use. 4.All negative and positive controls in the kit should be transferred to the sample preparation area and stored separately before the firstuse. 5. To prevent fluorescence interference, avoid touching the PCR reaction tube directly with bare hands, and avoid any marking on the PCR reaction tube. 6. The instrument amplification related parameters should be set in accordance with the relevant requirements of this manual, and differentbatchesof reagents cannotbemixed. 7.The product waste during the experiment should be detoxified before being discarded.
【Index of Symbol】
【FACTORY VIEW】
【FACTORY WORKSHOP】 【EXPORTER】
Magnus Internationa Limited
F12, New City Internationa Mansion A, 234 Huapao Ave. Liuyang, Hunan Province 410300 China
Contact: Goodwellmedical@gmail.com
【AUTHORIZED REPRESENTATIVE】
Lotus NL B.V. Koningin Julianaplein 10, 1e Verd, 2595AA, The Hague, Netherlands.
【PRODUCT AND STANDARDS】
【SHIPPING】 We are a trust healthcare and medical supplies from China. Be free to contact us at any time! You are very welcome to email us today at: Goodwellmedical@gmail.com for inquiry! We are exciting to build a reliable and long-term business with you at our best wishs! |
| Product Tags: MPV Nucleic Acid Detection Kit Monkeypox Virus MPV Detection Kit |